Electron transport chain
The electron transport chain (ETC, also called the respiratory chain) is a series of enzyme complexes embedded in the inner mitochondrial membrane through which electrons travel from nutrients all the way to oxygen. This process is responsible for producing most of the energy (ATP) in your body. Without a functional electron transport chain, you would lack the energy for a single heartbeat, a single thought, or a single breath.
- What the electron transport chain is and where it is located inside the mitochondrion
- Which four complexes make up the ETC and what each one does
- How ATP and deuterium-depleted water (DDW) are produced at the end of the chain
- Why cytochrome c oxidase (complex IV) is the key target for red light therapy
- What slows down or blocks the electron transport chain and how it relates to fatigue
- How light, grounding, and proper nutrition support ETC function
- What is the electron transport chain and where does it take place in the mitochondrion?
- Which four complexes make up the electron transport chain?
- How is ATP produced at the end of the electron transport chain?
- Why is metabolic water so important?
- Why is cytochrome c oxidase the key target for red light?
- What slows down or blocks the electron transport chain?
- How to support the function of the electron transport chain?
- How does the electron transport chain relate to fatigue and disease?

What is the electron transport chain and where does it take place in the mitochondrion?
The electron transport chain is a series of biochemical reactions in which electrons pass sequentially through four enzyme complexes (I, II, III, and IV) embedded in the inner membrane of mitochondria. Each electron transfer releases energy that is used to pump protons (H⁺) from the mitochondrial matrix into the intermembrane space.
The result is an electrochemical gradient, a kind of proton "pressure" that drives the enzyme ATP synthase. ATP synthase functions as a biological turbine, combining ADP with phosphate to produce ATP, the universal energy currency of every cell in your body.
The entire process is more formally known as oxidative phosphorylation (OXPHOS). It is the most efficient way the body extracts energy from food. While glycolysis (breaking down sugar without oxygen) produces only 2 ATP from a single glucose molecule, oxidative phosphorylation through the electron transport chain can generate up to 30 to 36 ATP from the same molecule.
Which four complexes make up the electron transport chain?
The electron transport chain consists of four main protein complexes, two mobile electron carriers (coenzyme Q and cytochrome c), and the enzyme ATP synthase (sometimes referred to as complex V). Each complex has a specific role:
Complex I (NADH dehydrogenase)
The largest complex in the entire chain. It accepts electrons from NADH (the reduced form of nicotinamide adenine dinucleotide), which is produced during the breakdown of fats, proteins, and carbohydrates. During electron transfer, it pumps 4 protons across the inner membrane. Complex I is also the primary site where reactive oxygen species (ROS), or free radicals, are generated when the chain is overloaded or damaged.
Complex II (succinate dehydrogenase)
The only complex that simultaneously serves as an enzyme of the Krebs cycle and the electron transport chain. It accepts electrons from succinate (an intermediate of the citric acid cycle) via FADH₂. Unlike the other complexes, it does not pump protons, so its energy contribution is smaller. Nevertheless, it is important because it provides an alternative entry point for electrons into the chain.
Complex III (cytochrome bc1 complex)
This complex accepts electrons from coenzyme Q (ubiquinone) and transfers them to cytochrome c. During this process, it pumps 4 protons across the membrane through the so-called Q cycle. Complex III is the critical "relay station" between the first and second halves of the chain.
Complex IV (cytochrome c oxidase, CCO)
The final complex and, from the perspective of photobiomodulation, the most important one. It accepts electrons from cytochrome c and transfers them to molecular oxygen (O₂). In the process, metabolic water (H₂O) is produced and 2 protons are pumped across the membrane. Cytochrome c oxidase is a chromophore, meaning it is a molecule capable of absorbing light. It is precisely this enzyme that red and near-infrared light targets during red light therapy.

How is ATP produced at the end of the electron transport chain?
Complexes I, III, and IV pump protons from the mitochondrial matrix into the intermembrane space. This creates a proton gradient, a difference in concentration and charge on both sides of the inner membrane. This gradient is a form of stored energy, much like water behind a dam.
When the "pressure" is great enough, protons flow back through the enzyme ATP synthase (complex V). ATP synthase is essentially a rotary molecular motor. It spins at speeds of up to 9,000 revolutions per minute, and with each rotation it combines 3 molecules of ADP with phosphate to produce 3 molecules of ATP.
Every day, your body produces an amount of ATP roughly equal to your own body weight. If you weigh 70 kg, your mitochondria synthesize and recycle approximately 70 kg of ATP daily. It is the most intensive biochemical process in the entire organism.
Why is metabolic water so important?
At the end of the electron transport chain, in complex IV, electrons combine with oxygen and protons to form water. This is not "ordinary" water. It is deuterium-depleted water (DDW), which has a significantly lower content of heavy hydrogen (deuterium) compared to the water we drink.
Deuterium is a heavy isotope of hydrogen. When too much of it accumulates inside the mitochondrion, ATP synthase slows down because deuterium is twice as heavy as regular hydrogen and "jams" the rotation of the molecular motor. That is why metabolic water from the electron transport chain is so valuable to mitochondria. It helps maintain low deuterium levels inside the cell.
From the Mitochondriak® philosophy perspective, the production of high-quality metabolic water is one of the key indicators of mitochondrial health. When the electron transport chain functions properly, sufficient DDW is produced and ATP synthase spins smoothly.
Why is cytochrome c oxidase the key target for red light?
Cytochrome c oxidase (CCO) in complex IV contains copper and iron centers that absorb light in the wavelength range of 600 to 1,000 nm, that is, in the red and near-infrared spectrum. This is precisely why this enzyme has become the cornerstone of all red light therapy (photobiomodulation).
Michael Hamblin from Harvard Medical School described the mechanism in his extensive review study (2018): red and NIR light is absorbed by CCO, leading to the dissociation of nitric oxide (NO) from the binding site on the enzyme. Under normal conditions, NO competitively inhibits CCO, effectively slowing it down. When light "detaches" NO, the enzyme runs at full capacity and the entire electron transport chain operates more efficiently (Hamblin, 2018).
The result is:
- Increased ATP production
- Increased production of metabolic water (DDW)
- A short-term rise in reactive oxygen species (ROS), which triggers an adaptive cellular response (hormesis)
- Release of nitric oxide, which dilates blood vessels and improves microcirculation
Lunova et al. (2020) confirmed that red light stimulates cytochrome c oxidase activity, while blue light inhibits it. This is one of the reasons why excessive blue light exposure from screens is problematic for mitochondrial function (Lunova et al., 2020).
What slows down or blocks the electron transport chain?
The electron transport chain is sensitive to many factors. When any of them disrupts its smooth operation, the result is reduced ATP production, increased free radical generation, and ultimately fatigue, inflammation, and slower recovery.
Excessive nitric oxide (NO)
NO is an important signaling gas, but in excess it blocks complex IV (CCO). This is one of the mechanisms through which chronic stress and inflammation lead to fatigue. Red light therapy helps remove this blockage.
High deuterium in food and water
Foods with high deuterium content (industrially processed sugars, tropical fruits out of season) increase the load on ATP synthase. The rotary motor slows down and energy production drops.
Artificial blue light after sunset
Blue light from screens and LED lighting inhibits CCO and disrupts the circadian rhythm, which indirectly weakens the entire electron transport chain. That is why it is important to minimize blue light in the evening and use, for example, blue light blocking glasses or red evening lighting.
Lack of oxygen and movement
Oxygen is the final electron acceptor in complex IV. Without an adequate oxygen supply, the entire chain shuts down. A sedentary lifestyle and shallow breathing reduce oxygen delivery to tissues.
Toxins and environmental stressors
Heavy metals (mercury, arsenic), pesticides, certain medications, and chronic oxidative stress damage the complexes of the electron transport chain. If the damage is long-term, mitochondria lose their ability to produce energy efficiently.
How to support the function of the electron transport chain?
From a mitohacking perspective, there are several proven ways to keep the electron transport chain in optimal condition:
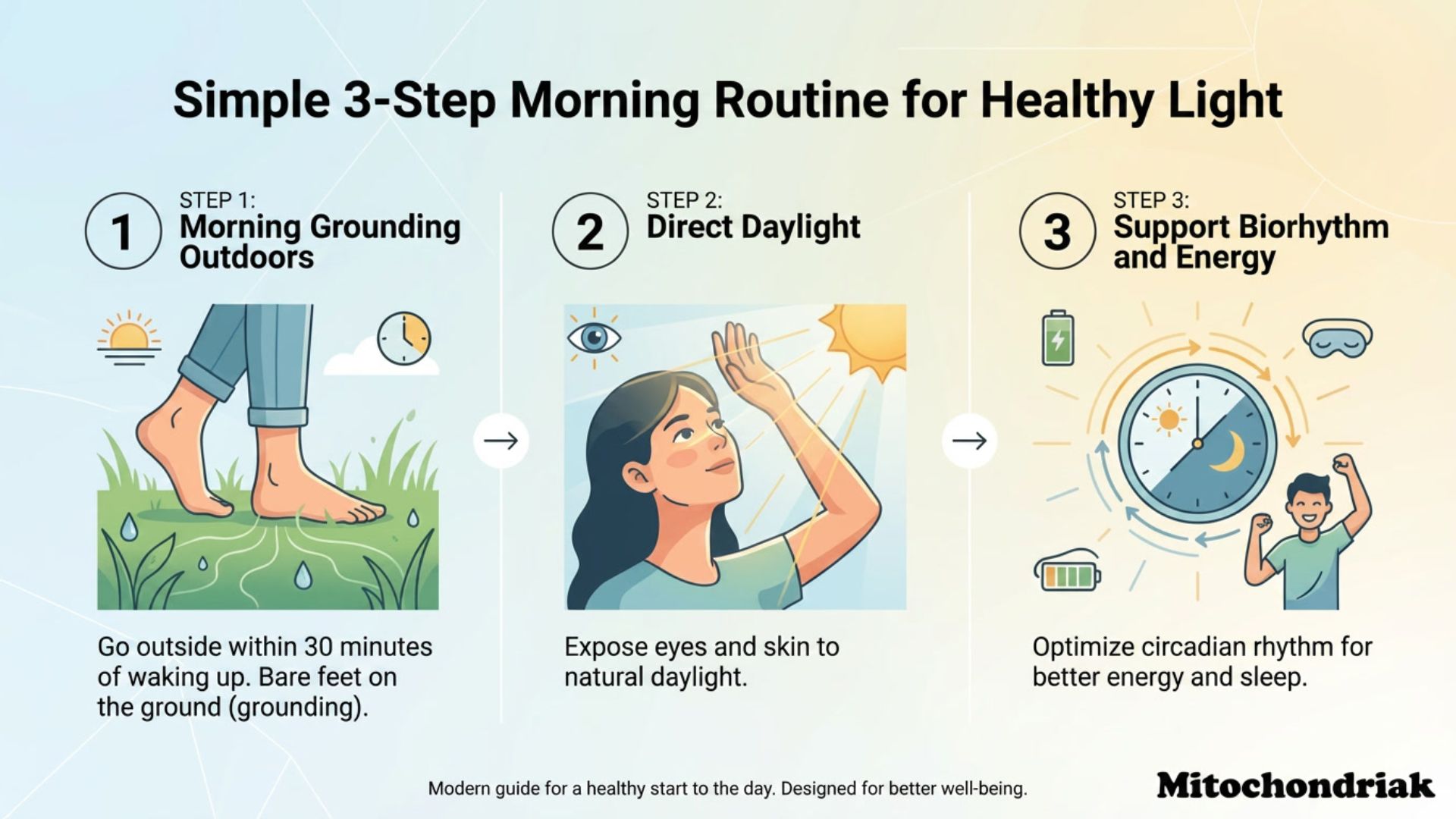
- Morning sunlight – contains red and infrared wavelengths that stimulate CCO and set the circadian rhythm of your mitochondria
- Red light therapy (photobiomodulation) – targeted application of wavelengths from 630 to 850 nm directly stimulates complex IV and increases ATP production
- Grounding (earthing) – supplies free electrons from the Earth that help neutralize excessive ROS generated in the chain
- Minimizing blue light in the evening – protects CCO from inhibition and supports nighttime production of melatonin, which is a powerful mitochondrial antioxidant
- High-quality, low-deuterium nutrition – seasonal, local foods, fatty fish, animal fats, and avoiding industrially processed sugars
- Movement and proper breathing – ensure an adequate supply of oxygen as the final electron acceptor
- Quality sleep in darkness – nighttime melatonin concentrates specifically in mitochondria, where it protects the electron transport chain from oxidative damage
How does the electron transport chain relate to fatigue and disease?
Dysfunction of the electron transport chain is a common denominator of many chronic diseases. When complexes I through IV do not work properly, the cell switches to a less efficient way of generating energy (aerobic glycolysis, the so-called Warburg effect). Less ATP is produced, more free radicals are generated, and the cell begins signaling stress.
Chronic fatigue, fibromyalgia, neurodegenerative diseases (Parkinson's, Alzheimer's), metabolic syndrome, and even premature aging all share a common foundation in impaired mitochondrial function and a dysfunctional electron transport chain. Kohler et al. (2023) emphasized in their review that the functional significance of electron transport chain supercomplexes extends beyond ATP production itself and reaches into the regulation of cell death, immune response, and epigenetics (Kohler et al., 2023).
This is precisely why supporting the electron transport chain is one of the pillars of mitohacking. It is not about a single supplement or a single habit. It is about creating an environment in which mitochondria can work the way evolution designed them to: with the right light, the right water, enough oxygen, and a minimum of toxic burden.
Related glossary terms
- Mitochondria – the cellular organelles where the electron transport chain resides
- ATP – the energy currency produced by the electron transport chain
- Photobiomodulation – light therapy that acts directly on complex IV of the electron transport chain
- Circadian rhythm – the biological rhythm that also governs the cyclical activity of mitochondria
- Melatonin – a mitochondrial antioxidant that protects the electron transport chain at night
Support your electron transport chain with red light
Mitochondriak® panels emit precise wavelengths (630, 660, 830, and 850 nm) that stimulate cytochrome c oxidase in your mitochondria. The result is more ATP, more metabolic water, and less oxidative stress. A session takes just 10 to 20 minutes per day.
- The electron transport chain is a system of 4 enzyme complexes on the inner mitochondrial membrane, where electrons pass from nutrients to oxygen
- Complexes I, III, and IV pump protons and create a gradient that drives ATP synthase
- At the end of the chain, ATP (energy), metabolic water (DDW), and heat are produced
- Cytochrome c oxidase (complex IV) absorbs red and NIR light, which is the basis of photobiomodulation
- Excessive blue light, nitric oxide, deuterium, and toxins slow down or block the electron transport chain
- Morning sunlight, red light therapy, grounding, and quality sleep are the most effective ways to support the ETC
Sources and References
- Hamblin, M. R. (2018). Mechanisms and Mitochondrial Redox Signaling in Photobiomodulation. Photochemistry and Photobiology, 94(2), 199–212. PMC5844808
- Ahmad, M., Wolberg, A., Kahwaji, C. I. (2023). Biochemistry, Electron Transport Chain. StatPearls. NBK526105
- Lunova, M. et al. (2020). Light-induced modulation of the mitochondrial respiratory chain activity: possibilities and limitations. Cellular and Molecular Life Sciences, 77(14), 2815–2838. PubMed 31583425
- Wang, X., Tian, F., Soni, S. S. et al. (2016). Interplay between up-regulation of cytochrome-c-oxidase and hemoglobin oxygenation induced by near-infrared laser. Scientific Reports, 6, 30540. PMC4971496
- Kohler, A. et al. (2023). The functional significance of mitochondrial respiratory chain supercomplexes. EMBO Reports, 24(11), e57092. PMC10626428
- Poyton, R. O., Ball, K. A. (2011). Therapeutic photobiomodulation: nitric oxide and a novel function of mitochondrial cytochrome c oxidase. Discovery Medicine, 11(57), 154–159. PubMed 21356170

.png)